|
This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". 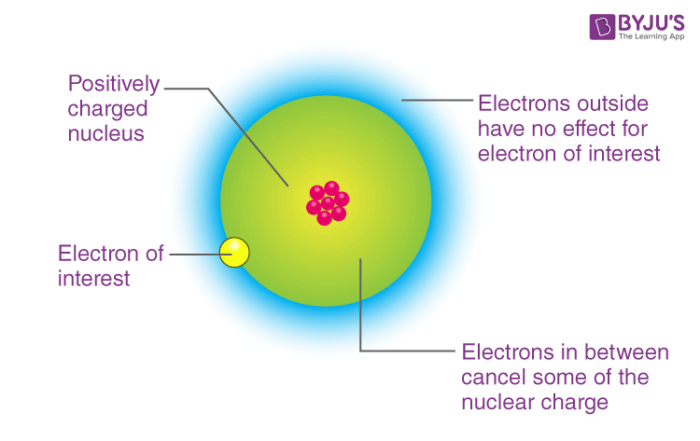
The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. Since electrons are about 2000 times less massive than protons (and neutrons), then the mass of an atom is mostly from the protons and neutrons. If by “biggest” and “smallest”, you mean mass (which is a measure of how much matter is there), then the smallest is the hydrogen atom with one proton and one electron. Silicon’s outer level feels a weaker charge because of +4 and therefore it is not pulled as close to the nucleus as sulfur’s outer level. Using the concepts of shielding and attraction explain why sulfur is smaller in radius than silicon. Does sulfur have a larger atomic radius than silicon? Phosphorus has a larger atomic radius than Sulfur. Which element has a larger atomic radius than sulfur? So it is highest in it’s period and highest in the group. Which of the following ions has largest size?Ĭs+ has highest ionic radii as it is on the extreme right corner of the table. Magnesium nucleus will attract the electrons more toward it and therefore, shrinking the size of the atom. Magnesium atom is smaller than sodium atom because the nuclear charge of Magnesium ( 12+ ) is higher than that of Sodium ( 11+ ). 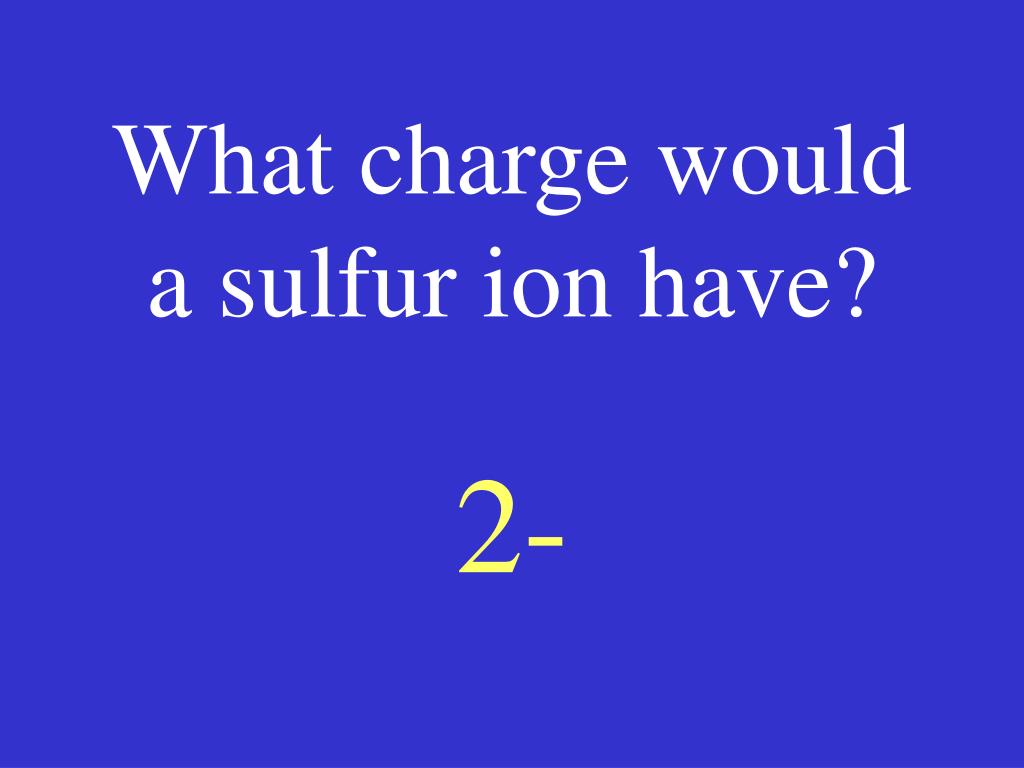
Hydrogen is the smallest atom of all as it is having just one electron. Which of the following atoms will have the smallest size? An electron gets added to each element from left to right and in the same shell.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
